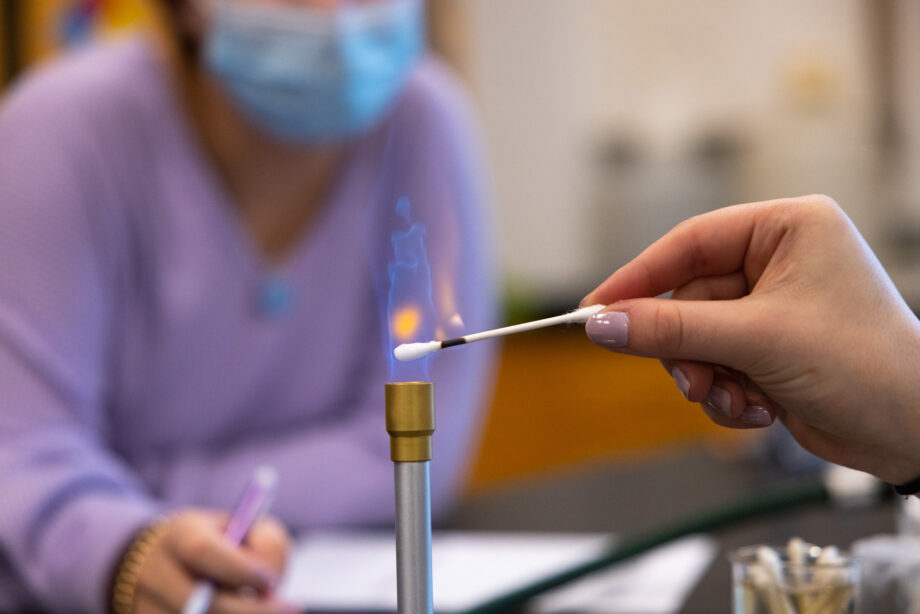
Snow blankets the fields outside the windows of Baker 211, but inside things are heating up as students huddle around lab tables in groups, preparing their Bunsen burners to add some color to the gray day with the much-anticipated flame tests. When the late February doldrums hit, Chemistry class with Dr. Burke is where you want to be.
“In Chemistry class,” says Burke, “the focus is on studying matter, its properties, and the changes it undergoes; working in the lab and having a hands-on experience of how the concepts we are covering govern the larger world is such a critical part of science education.” Her students have covered nuclear properties in their previous unit and are now learning about the quantum mechanical model of the atom and about their internal structure in order to understand how atoms combine to form compounds. Eager to light the Bunsen burners, students listen to Burke’s overview of the lab and safety precautions, excited to see the results.
“When elements are heated to high temperatures,” explains Burke, “the electrons within the atoms of the element may be promoted to higher energy levels. When these high-energy electrons return to their ground state, the absorbed energy is released in the form of photons or particles of light. If those photons have wavelengths in the visible range, we will perceive them as a color. A straightforward way of determining if an element will produce photons in the visible range or not is to conduct a flame test.”
In their small groups, students use cotton swabs to take samples of various solutions and heat them in the Bunsen burners; by observing colors and comparing results they are able to identify the element responsible for producing each color and identify unknown samples by the color they produced. Students gasp, noting the outcomes and celebrating their predictions as flames turn blue, red, green and orange. Burke explains that the flame test lab is not only a straightforward way of determining whether an element will produce photons in the visible range, but also serves as a way for chemistry students “to relate what is happening on the atomic level to what we can observe macroscopically. We can explain something too small for us to observe—the energy state of electrons—by observing something we are more familiar with: light and color.”
Just another day in the Baker science labs!